 
Why is ClF3 T-shaped and not trigonal planar?Ĭhlorine trifluoride has 5 regions of electron density around the central chlorine atom (3 bonds and 2 lone pairs). There is also an asymmetric charge distribution around the central atom. It acquires such shape because of the presence of two lone pairs which take up equatorial positions and there are greater repulsions. If the central atoms contain 5 bond repulsion units and if it doesn’t contain a lone pair on the central atom, the molecule shape is trigonal bipyramidal having a bond angle of 175° F-Cl-F.ĬlF3 molecular geometry is said to be a T-shaped. What is the shape and geometry of ClF3?Ĭhlorine Trifluoride (ClF3) represents a Trigonal bipyramidal geometry. That leaves one equatorial, and 2 axial, sites where the Cl-F bonds can go, which leaves you with a T-shaped geometry, rather than a trigonal planar structure. Therefore, the interactions are lowest in the equatorial environment, so this is where the lone pairs go. H2 CO is polar because the shared electrons between the carbon atom and the oxygen atom are not shared equally between the two. According to the AX3N2 formula, ClF3 molecular geometry is T-shaped and electron geometry is trigonal pyramidal. With the help of the ClF3 Lewis dot structure, we know chlorine is the central atom that contains 2 lone pairs and is attached to 3 bonded atoms. What is the polarity of ClF3?ĬlF3 is a polar molecule because it has an asymmetrical shape and the presence of 2 lone pair electrons leading to an unequal distribution of charge making this molecule polar in nature.ĭoes ClF3 have trigonal pyramidal geometry? The electron geometry of chlorine trifluoride is trigonal bipyramidal with a 175° F-Cl-F bond angle. 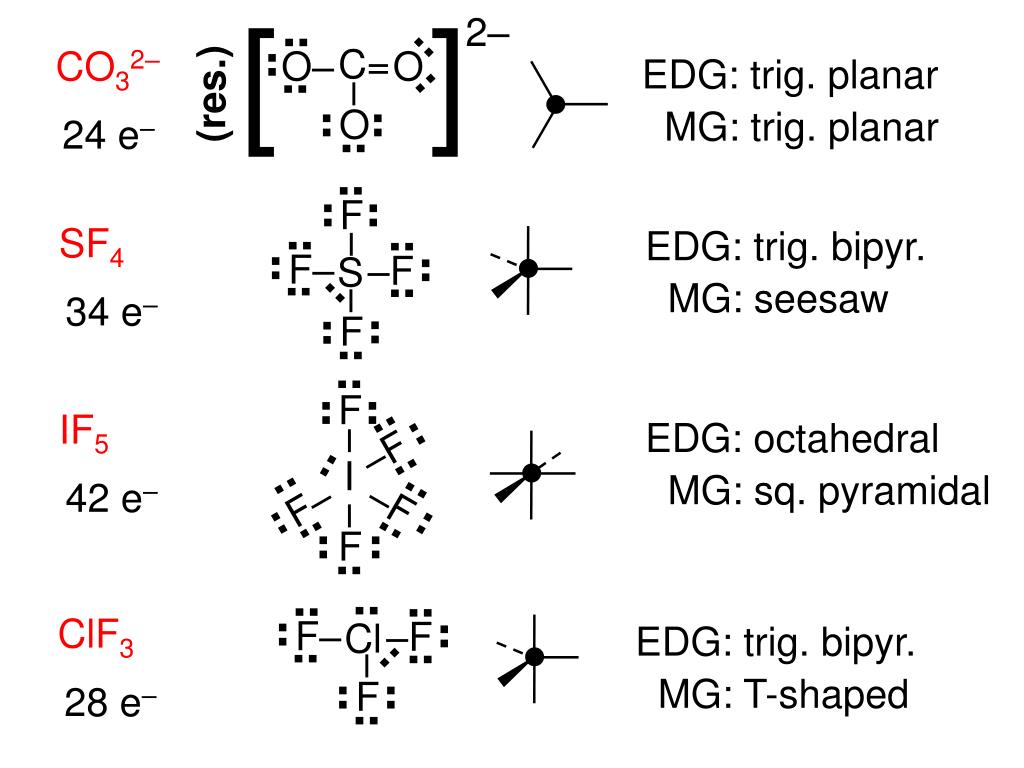
Is ClF3 a geometry?ĬlF3 molecular geometry is said to be a T-shaped. Solved For ClF3, the molecular geometry is Trigonal |. What is the molecular geometry of ClF3 chegg? 7 What is the electron-pair geometry of ClF3?.6 What is the shape and geometry of ClF3?. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
